- Home
- Services
- About
- News
- Contact
- Sentience nioh 2
- Best program language to learn first for website
- Pro paint bare study
- Best dwarf fortress tileset for noobs
- Finicky used furniture prescott valley
- Ver betty la fea
- Supply demand semaphor indicator
- Glam furniture
- Chambers dictionary online free amazon
- As element periodic table
- Mano to irani songs
- Program to print envelopes free
- Microsoft word for mac 2018
- Fernbus simulator pc
- Stem cells definition

The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. Look for the Sign in button at the top of each page.A vertical column in the periodic table.
#AS ELEMENT PERIODIC TABLE FREE#
Registering an account for the Science Learning Hubs is easy and free – sign up with your email address or Google account. You can then add additional content, notes and make other changes. Login to make this collection part of your private collection, just click on the copy icon. The Science Learning Hub team has curated a collection of resources related to the periodic table of elements. Symbol find – In this activity, students become familiar with symbols of the chemical elements by creating them using letters from a phrase or sentence.Element rap – In this activity, students become familiar with the names and symbols of the chemical elements by creating a rap or poem.Atomic clock – Use the atomic clock teacher resource to familiarise students with the names and symbols of the chemical elements.Development of the periodic table – Use this article to find out about the first scientific discovery of an element in 1649 and how this grew into the periodic table as we know it today.It has the same number of electrons in its outermost energy level as the other elements in Group 1, but as it is a gas, it is normally put in the non-metal group. Hydrogen has the atomic number 1, so it sits at the top left of the table with the alkali metals of Group 1. Metals, semi-metals and non-metals – These share some properties, but not enough for a group to fill a column. The atomic numbers of these elements actually sit in the bottom left corner of the main table. Transition metals – Periodic tables have an extra block of transition metals at the bottom, for elements called rare-earths (or lanthanides) and actinides. The groups of elements with similar properties have names and are normally coloured differently in a periodic table. The more you find out about atomic structure, the more patterns and relationships you’ll find in the periodic table. Other patterns are there, too, For example, the energy needed to get an electron away from an atom: Atomic number increases as you move down a column.Atomic number increases as you move right along a row.You’ll notice that elements in the periodic table are arranged in rows and columns.Įlements in a group (column) have the same number of outer electrons, so they have similar chemical properties. For example, the square for iron will look a bit like this: In some versions of the table, these squares can contain a lot of writing, but to start with, three pieces of information are enough – each square should contain the name of the element, its official chemical symbol and its atomic number.
#AS ELEMENT PERIODIC TABLE FULL#
It will help to have a copy of the periodic table – the one here will get you started, or you can print a full one from the website below.Įach element has a square in the periodic table. The element iron is made only of iron atoms, and iron atoms are the same everywhere – iron atoms on Earth are the same as iron atoms on Mars. Elements cannot be split into simpler substances using normal chemical methods. ElementsĪn element is a substance that consists of atoms with the same atomic number. From this, we can work out how the electrons are arranged, and this will tell us how an element will react with others. The atomic number can also tell us how many electrons an atom has.
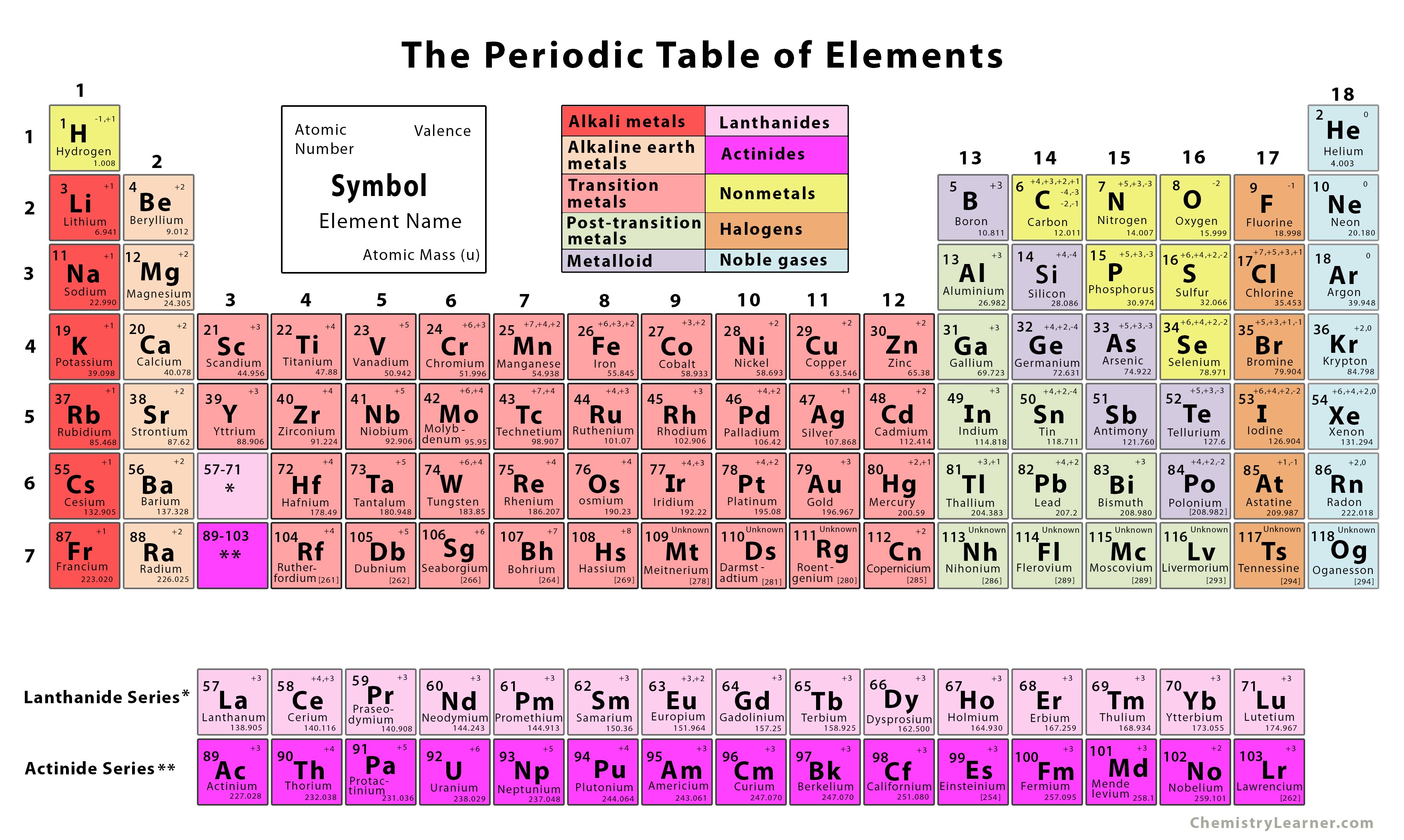
For example, if an atom has six protons, it can only be carbon. The atomic number of an element is the number of protons an atom has – the number of protons determines what an element is. The electrons are organised in energy levels – how an element behaves depends on how easy it is to gain or lose electrons in the outermost energy levels. Electrons move around within a large area of space outside the nucleus. Protons and neutrons form the nucleus at the centre of an atom (hydrogen is a bit different, it only has a proton). There are smaller particles, but they don’t concern us here. An atom has three basic parts – protons, neutrons and electrons.
- Home
- Services
- About
- News
- Contact
- Sentience nioh 2
- Best program language to learn first for website
- Pro paint bare study
- Best dwarf fortress tileset for noobs
- Finicky used furniture prescott valley
- Ver betty la fea
- Supply demand semaphor indicator
- Glam furniture
- Chambers dictionary online free amazon
- As element periodic table
- Mano to irani songs
- Program to print envelopes free
- Microsoft word for mac 2018
- Fernbus simulator pc
- Stem cells definition
